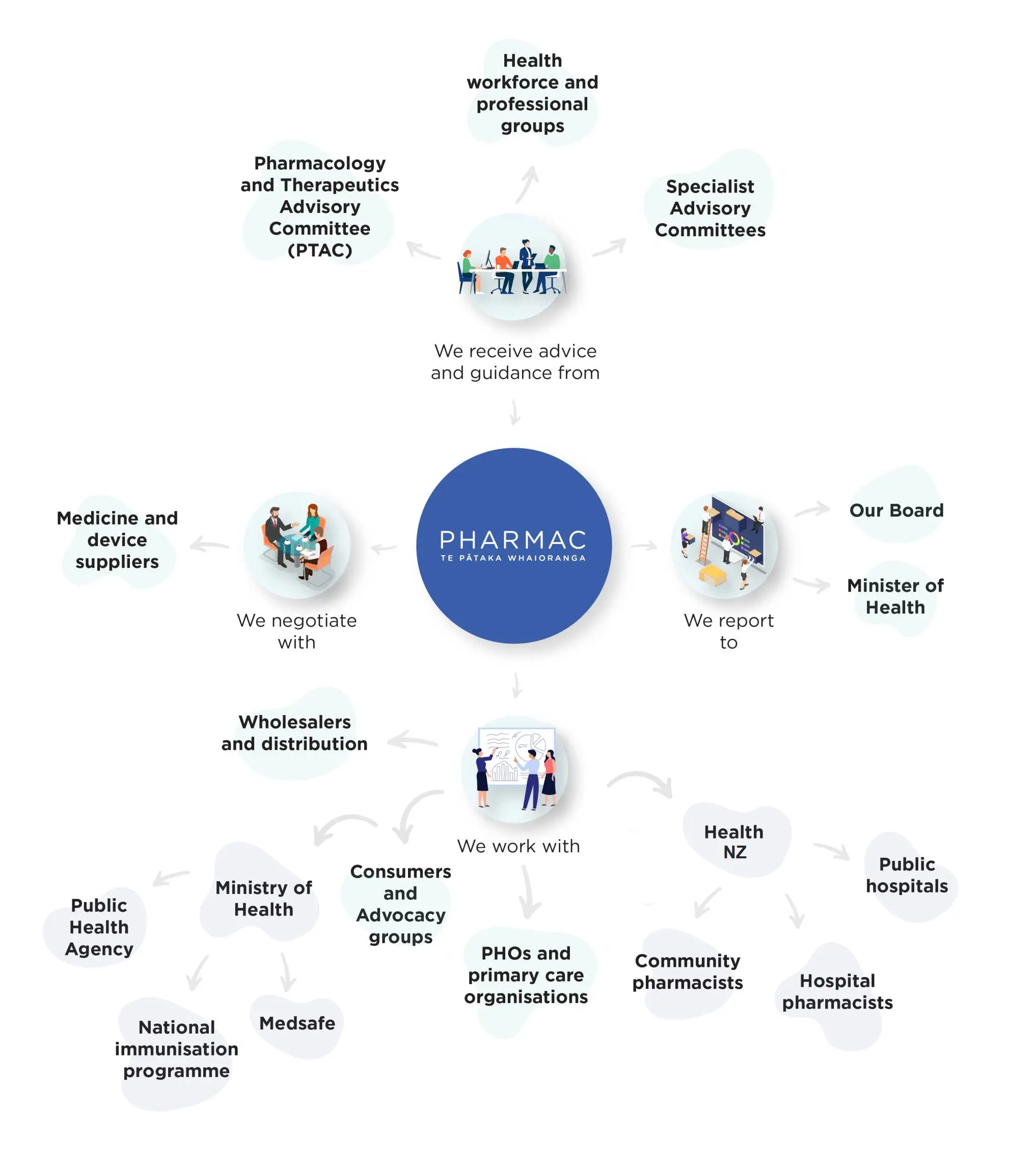
Our place in the health system
Pharmac’s role within the New Zealand health system is to make decisions on which medicines and related products are funded for the best health outcomes from within the available funding. We also contract for hospital medical devices.
Our relationships and accountabilities
We interact with a wide range of stakeholders in our work.
Minister of Health
The Minister takes responsibility for Pharmac's performance. The Minister and government of the day set Pharmac's budget.
The expectations of Pharmac are set by the Minister who is also involved in appointing the Pharmac Board.
The Pharmac Board provide reports and briefings regularly to the Minister of Health.
Parliament and Members of Parliament
As a Crown entity, Parliament holds us to account for our actions. We help the Minister respond to Parliamentary Questions (questions asked about our operations by Members of Parliament). Our performance is also scrutinised by Parliament’s Health Select Committee. Pharmac also helps MPs with queries from their local constituents.
Health Select Committee on parliament.nz(external link)
Health New Zealand | Te Whatu Ora
Health NZ holds the funding to deliver most publicly financed health services, including public hospitals and the medical devices they use. We work closely with clinicians and procurement staff when making decisions about hospital medical devices.
Sector Operations Group
Sector Operations Group is Health NZ’s claims processing unit. It pays pharmacists for the subsidised medicines people get from their community pharmacy. It also processes Special Authority applications for medicines with specific criteria that must be met before they will be funded. Pharmac decides what the criteria are and Health NZ checks that applicants meet the criteria.
Ministry of Health
We work with the Ministry of Health, which acts on behalf of the Minister, to track Pharmac’s performance.
We engage with the Ministry on a range of issues, including policy development. We have close working relationships with several of the Ministry’s business units, including Medsafe.
Medsafe
Medsafe decides which medicines, related products, and medical devices are safe and effective for New Zealanders to use. They check that these products meet standards of quality, safety and efficacy. We usually consider a medicine or related product for funding only after approval by Medsafe (but there can be good reasons for funding an unapproved medicine).
CARM
The Centre for Adverse Reactions Monitoring (CARM) in Dunedin is New Zealand’s national monitoring centre for adverse reactions. These include the side effects of a medication or surgery. It collects and evaluates reports of adverse reactions to medicines, vaccines, herbal products and dietary supplements from health professionals in New Zealand. Reports are then provided to Medsafe.
Report an adverse reaction(external link)
Clinicians and other prescribers
Prescribers include general practitioners, specialists, and nurses. They put the Pharmaceutical Schedule into practice. They refer to it on a regular basis to determine which subsidised medicines are available for their patients.
We have a network of about 160 clinical experts we regularly seek advice from for a range of decisions we make. We also seek advice from any clinician when we consult on potential funding decisions.
Pharmacists
In the community, pharmacists dispense prescriptions and advise people on how and when to use the prescribed medicine. In the hospital, pharmacists play a vital role in ensuring the funded treatments are available as prescribed. As with prescribers, we rely on feedback from pharmacists about the practicality of our decisions.
Health professional groups
Professional associations for doctors, pharmacists, nurses, and other health professionals (such as, clinical engineers) provide an important perspective on medicine funding issues and our approach to hospital medical devices management. We meet with such groups and seek their input through our consultation processes. Working alongside these organisations, we develop activities to manage changes in medicines, improve access to and encourage responsible use of pharmaceuticals.
Consumer and patient groups
The decisions Pharmac makes affect almost all New Zealanders, so it is important that the impact of those decisions on people is well thought through. We are in regular contact with patient and consumer groups and welcome feedback on medicine funding, our devices work, or other issues.
We also have a Consumer Advisory Committee who we seek advice from on a range of issues.
Meet the Consumer Advisory Committee
Medicine and device suppliers
We rely on suppliers to provide medicines and medical devices for New Zealanders. In most cases, when we fund a medicine we agree on a supply contract with the company to ensure they continue to supply the contracted medicines. We also agree contracts for the supply of hospital medical devices, they are still funded by Healht NZ hospitals.
Before a product is listed in the Schedule, suppliers must provide information on:
- how a medicine or medical device works
- its interactions and side effects
- the information we need to assess the product for listing.
Once listed, suppliers need to tell us if:
- there is any change to the medicine or medical device
- if the price is going to change
- if there's a supply issue
- whether a product is going to be discontinued


