Decision for funded multiple sclerosis treatments
What we’re doing
We're pleased to announce some changes to the way that multiple sclerosis treatments are funded, as well as widening access to the funded treatments, from 1 March 2021. In summary:
- access to funded multiple sclerosis treatments will be widened for people with an Expanded Disability Status Scale (EDSS) score of 0 to 6.0 (inclusive) meaning treatment will continue to be funded for patients while their EDSS is 6.0 or below.
- applications to the Multiple Sclerosis Treatment Assessment Committee (MSTAC) will no longer be required and funded multiple sclerosis treatments will be accessed by a standard Special Authority application in the same way as many other funded medicines.
The funded multiple sclerosis treatments are natalizumab (Tysabri), fingolimod (Gilenya), the beta interferons (interferon beta-1-beta (Betaferon), interferon beta-1-alpha (Avonex)), glatiramer acetate (Copaxone), teriflunomide (Aubagio), dimethyl fumarate (Tecfidera) and ocrelizumab (Ocrevus).
Further details of this decision, including the feedback we received on the Consultation and background information can be found on the following pages.
Any changes to the original proposal?
This decision was subject to a consultation letter dated 27 November 2020.
We thank all those who provided consultation feedback. Following review of the feedback received we have made some changes to the Special Authority criteria we consulted on. The consultation responses we received and our response to the points raised are summarised below.
Who we think will be most interested
- People with multiple sclerosis, and their whānau, family, and caregivers
- Neurologists, neurology nurses, general physicians, general practitioners, and other health professionals involved in the management and treatment of multiple sclerosis
- Health groups with an interest in multiple sclerosis
- Pharmacies and DHBs
- Pharmaceutical suppliers
Detail about this decision
From 1 March 2021 we will be implementing the following:
- We are widening the funding eligibility criteria to people with multiple sclerosis who have an Expanded Disability Status Scale (EDSS) score of 0 to 6.0 (inclusive). An EDSS of 6.0 means that a person can walk 100 metres with or without rest and/or assistance (including a cane, crutch, or brace). The EDSS is a method of quantifying disability in multiple sclerosis and is used to measure and assess disease progression.
- The same entry criteria for access to funded treatment will apply to all of the currently funded multiple sclerosis treatments. Previously, there were different sets of criteria that restricted switching between the different multiple sclerosis treatments.
- The process for obtaining funded access to all multiple sclerosis treatments is changing to a standard Special Authority. Applications to the Multiple Sclerosis Treatment Assessment Committee (MSTAC) are no longer required. This will save time for prescribers because they will be able to apply for funding online and get an immediate decision through the Electronic Special Authority system.
- People with a current funding approval via MSTAC will have their approvals transferred to a standard Special Authority; no additional action by clinicians will be required.
- There are no changes to the current eight funded multiple sclerosis treatments. They are: natalizumab (Tysabri), fingolimod (Gilenya), the beta interferons (interferon beta-1-beta (Betaferon), interferon beta-1-alpha (Avonex)), glatiramer acetate (Copaxone), teriflunomide (Aubagio), dimethyl fumarate (Tecfidera), and ocrelizumab (Ocrevus).
Information for people with multiple sclerosis
- You will continue to receive your funded multiple sclerosis treatment in the same way as you do now. There will be no changes to the cost you currently pay for your treatment or the way you access your treatment.
- Most people with clinically definite, relapsing-remitting multiple sclerosis (CD RR-MS) will be able to receive their funded treatment for longer.
- The way your doctor or specialist applies for funding on your behalf is changing. Applications for funded multiple sclerosis treatments will be processed differently after 1 March 2021.
- People with multiple sclerosis who stopped treatment due to no longer meeting the previous eligibility criteria may now be eligible to restart funded treatment under the new criteria. People should talk to their specialist to determine if they meet the new renewal criteria.
- If you do not meet the renewal eligibility criteria due to a temporary non-multiple sclerosis related reason, your doctor or specialist will be able to apply for a Special Authority waiver for an extension of six months.
- PHARMAC anticipates all people with multiple sclerosis who are currently receiving funded treatment to be affected positively by this change.
Information for prescribers
- The way applications for multiple sclerosis treatments are processed is changing on 1 March 2021. Instead of applications being considered by PHARMAC’s group of experts, MSTAC, they will be processed by the Ministry of Health in the same way that other Special Authorities are processed - through the electronic Special Authority (ESA) system.
- Neurologists and general physicians are eligible to apply for funded multiple sclerosis treatments. Neurologists and general physicians will continue to be able to delegate the writing of prescriptions for people with multiple sclerosis to any prescriber (including the person’s GP).
- It will be less burdensome for clinicians to apply for funded MS treatments, and by using the standard Special Authority system decisions will be available immediately.
- Clinicians do not need to take any action for people who are already accessing funded multiple sclerosis treatments. Current Special Authority approvals will be transferred to the standard Special Authority system with the same expiry dates. Those due to expire at the end of February 2021 and end of March 2021 will be extended by one month to support a smooth transition.
- People who have current approvals for funded multiple sclerosis treatment before 1 March 2021 will be automatically transitioned to a new approval and Special Authority number.
- Sector Operations Group at the Ministry of Health will not send out letters with the new approval numbers. If you need the new numbers for your records you can look them up on the ESA system or call them on 0800 243 666, option 3 and then option 2.
- There are minor changes to the eligibility criteria. The intention of these changes is to ensure that people with multiple sclerosis who are currently accessing funded treatment are not disadvantaged by the new criteria, while ensuring that funding is made available to those new people that are eligible for treatment.
- The new Special Authority criteria will:
- Widen access of currently funded multiple sclerosis treatments to an EDSS of 0-6.0 (inclusive).
- Ensure patients who started treatment under older eligibility criteria are not disadvantaged.
- Generate one standard set of Special Authority criteria for all currently funded multiple sclerosis treatments.
- Remove criteria that are overly prescriptive and leave certain clinical decisions to be made by clinicians acting within their scope of practice, particularly around which funded MS treatment patients can access.
- Prescribers who have patients who may not meet the access criteria due to a temporary non-MS related reason can apply for a Special Authority waiver for an extension of six months.
- The small number of patients with current approvals under older eligibility criteria that allow them to stay on treatment above EDSS of 6.0 will be ‘grand-parented’ and will be granted special approvals by PHARMAC to ensure that they will be able to remain on their current treatments with their current stopping criteria. This applies to 10 patients. We will communicate directly with the clinicians treating these 10 patients to inform them of the process to apply for an annual renewal.
Information for pharmacists
- Pharmacists will need to update their patient’s Special Authority number when dispensing their funded multiple sclerosis treatment.
- Sector Operations Group at the Ministry of Health will not send out letters with the new approval numbers. If you need the new numbers you can call them on 0800 243 666, option 3 and then option 2.
- Pharmacists will be reimbursed in the same way as they are reimbursed for all other treatments funded via Special Authority.
- The way applications for multiple sclerosis treatments are process is changing on 1 March 2021. Instead of applications being considered by MSTAC, they will be processed electronically by the Ministry of Health in the same way that other Special Authorities are processed.
New Special Authority criteria
From 1 March 2021, funded access to the eight funded MS treatments will be amended in Section B (the Community) of the Pharmaceutical Schedule. Equivalent changes will also be made in Part II of Section H (the Hospital Medicines List) of the Pharmaceutical Schedule. The new criteria will be:
Special Authority for Subsidy
Initial application – (Multiple sclerosis) from a neurologist or general physician.
Approvals valid for 12 months for applications meeting the following criteria:
All of the following:
- Diagnosis of multiple sclerosis (MS) must be confirmed by a neurologist. Diagnosis must include MRI confirmation; and
- Patients must have Clinically Definite Relapsing multiple sclerosis with or without underlying progression; and
- Patients must have an EDSS score between 0 – 6.0; and
- Patient has had at least 1 significant relapse of multiple sclerosis in the previous 12 months or 2 significant relapses in the past 24 months; and
- All of the following:
- Each significant relapse must be confirmed by the applying neurologist or general physician (the patient may not necessarily have been seen by them during the relapse but the neurologist/physician must be satisfied that the clinical features were characteristic); and
- Each significant relapse is associated with characteristic new symptom(s)/sign(s) or substantially worsening of previously experienced symptoms(s)/sign(s); and
- Each significant relapse has lasted at least one week; and
- Each significant relapse has started at least one month after the onset of a previous relapse; and
- Each significant relapse can be distinguished from the effects of general fatigue; and is not associated with a fever (T> 37.5°C); and
- Either
- Each significant relapse is severe enough to change either the EDSS or at least one of the Kurtze Functional System scores by at least 1 point; or
- Each significant relapse is a recurrent paroxysmal symptom of multiple sclerosis (tonic seizures/spasms, trigeminal neuralgia, Lhermitte’s symptom); and
- Evidence of new inflammatory activity on an MR scan within the past 24 months; and
- Any of the following:
- A sign of that new inflammatory activity is a gadolinium enhancing lesion; or
- A sign of that new inflammatory activity is a lesion showing diffusion restriction; or
- A sign of that new inflammatory is a T2 lesion with associated local swelling; or
- A sign of that new inflammatory activity is a prominent T2 lesion that clearly is responsible for the clinical features of a recent relapse that occurred within the last 2 years; or
- A sign of that new inflammatory activity is new T2 lesions compared with a previous MR scan.
Note: Natalizumab can only be dispensed from a pharmacy registered in the Tysabri Australasian Prescribing Programme operated by the supplier. Treatment on two funded multiple sclerosis treatments simultaneously is not permitted.
Renewal– (Multiple sclerosis) only from a neurologist or general physician. Approvals valid for 12 months where patient has had an EDSS score of 0 to 6.0 (inclusive) at any time in the last six months (i.e. the patient has walked 100 metres or more with or without aids in the last six months).
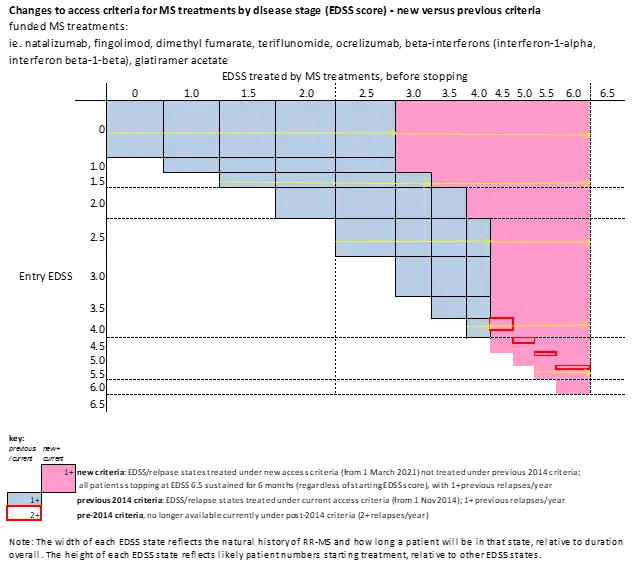
Schematically, these changes can be depicted in the following diagram:
Our response to what you told us
We’re really grateful for the time people took to respond to this consultation. A summary of the main themes raised in feedback, our responses to the feedback received, and changes we have made after listening to you are available on our notification webpage.
Summaries of what PHARMAC staff believe are the significant matters raised in these responses are provided below.
|
Theme |
PHARMAC Comment |
|
Responders were generally supportive of the proposal to widen access and to transition to a standard Special Authority |
This was consistent with our view; we believe this decision will result in positive outcomes for patients. |
|
Requests to change the proposed Special Authority criteria, including:
|
The intent of the Special Authority criteria is to ensure that people with multiple sclerosis who are currently accessing funded treatment are not disadvantaged by the new criteria, and align with current requirements; while also ensuring that funding is made available to those new people that are eligible for treatment. Some small changes have been made to the wording of the Special Authority criteria following careful consideration of feedback, including the removal of ‘remitting’ from the criteria. |
|
The requirement for both a clear clinical diagnosis of multiple sclerosis and MRI evidence is a barrier to treatment as timely access to an MRI is difficult and varies across DHBs. |
PHARMAC acknowledges timely access to MRI may be difficult and varies across DHBs. MRI is only required for the initial criteria (as is currently required); MRI is not required for ongoing treatment (ie. as part of the renewal criteria). |
|
Noted that the proposal may lead to a modest increase in MRI scans and clinic workload. The proposal may allow stable people with multiple sclerosis to be assessed by health professionals other than neurologists which would modestly reduce clinic visits. |
We appreciate that this change may impact resource for the health system, and the additional costs for infusion services have been considered. Special Authorities must be made by neurologists and general physicians. However, they would continue to be able to delegate the writing of prescriptions for people with MS to the person’s GP. |
|
Requested detail on how the Special Authority process would work in practice and the process for resolving ‘disagreements’ regarding funded MS treatment decisions made under Special Authority, including where people with multiple sclerosis who may not meet the criteria due to a non-multiple sclerosis related reason. |
The Special Authority for MS treatments will operate in the same way as all other Special Authority forms. As with other Special Authorities, waivers are available for patients who meet the intent of the Special Authority criteria. |
|
Noted the importance of ensuring people with multiple sclerosis who have ceased treatment due to previous criteria and would now be eligible under the proposed criteria are aware.
|
PHARMAC will communicate with relevant health professionals and stakeholder groups and will highlight that people with multiple sclerosis who have ceased treatment due to previous exit criteria may now be eligible for funded treatment under the new renewal criteria. |
|
Request for clarification on how people with MS can access funded medications when returning from overseas or move to New Zealand. Concern particularly for those who are on treatment due to wider access overseas and would not meet the NZ criteria for access.
|
Patients and their prescribers will be required to show that they met or would have met the New Zealand criteria for access prior to commencing treatment overseas. This requirement applies to all treatments funded with SA criteria in New Zealand, including the current criteria for access to funded MS treatments. There is no proposed change to this process. |
|
Request to clarify that the 100 m walk can be sighted and approved by a health professional other than a neurologist, eg. GP or physiotherapist. |
Special Authority renewals must be applied for by a neurologist or general physician. How the criteria are assessed is at the discretion of the clinician, as is the case now. |
|
Noted that there would be no opportunity to critically review the diagnosis of multiple sclerosis ie. via MRI reports etc. |
PHARMAC has accounted for the lack of review of MRI reports in setting the proposed Special Authority criteria and the estimates of patient numbers likely to access funding through this proposal. This remains the same as is the case under current access. |
|
A concern was raised regarding how people with MS receive their infusion treatment, eg. some people with multiple sclerosis are required to collect their infusion treatment from a community pharmacy and transport it to their infusion clinic. |
There are no proposed changes to the way that patients access their multiple sclerosis treatments as part of this proposal.
|
|
Queried if annual prescriptions could return, noting the increased effort to renew frequently through a GP. |
Prescription frequency for all pharmaceuticals in New Zealand is three-monthly (with the exception of oral contraceptives). This frequency is a legal requirement as per Medicines Regulations 1984 (section 39A(external link)). There are no changes proposed to the frequency of prescriptions as part of this proposal. |
|
Requests for further widened access to multiple sclerosis treatments, including later stopping criteria (ie. higher EDSS), and the removal of the requirement for relapse. |
We would welcome funding applications for relevant changes to funding criteria. More information about how to make a funding application can be found here. |
Who to contact
If you have any questions about this decision, you can email us at enquiry@pharmac.govt.nz; or call our toll free number (9 am to 5 pm, Monday to Friday) on 0800 660 050.


