Decision to simplify access for antiviral COVID-19 treatments
What we’re doing
We're pleased to announce that we have made the decision to simplify access to three antiviral medicines used to treat early COVID-19 from 14 September 2022:
- nirmatrelvir with ritonavir (supplied under the brand name Paxlovid)
- molnupiravir (supplied under the brand name Lagevrio) and
- remdesivir (supplied under the brand name Veklury)
The changes mean that more New Zealanders who get COVID-19 will be able to access these treatments.
Criteria changes come into effect on Wednesday 14 September 2022 and will make these treatments available to a larger priority population of people with early COVID-19 at risk of severe illness. This update expands on previous changes, simplifies the criteria and more than doubles the size of the eligible population.
The additional groups that will now be eligible for treatment include:
- Anyone 65 years age or over
- Anyone of Māori or Pacific ethnicity who is 50 years age or over
- Anyone 50 years age or over who hasn’t completed their primary course of vaccination (at least two doses)
- Anyone else with three or more high-risk medical conditions.
Our original Access Criteria that identified people at highest risk of severe illness were developed based on advice from our clinical expert advisors and other stakeholders. Over time we have updated the criteria, most recently in July 2022, to address new information about antiviral access rates, New Zealand populations impacted more severely by COVID-19 and local rates of COVID-19 hospital admissions.
With the reducing COVID-19 case numbers and increased confidence in the antiviral supply available, we can support this change to make the criteria simpler while ensuring that those people at risk are eligible for treatment. We expect this may remove some barriers to access of funded treatment and make it easier for health professionals to consider treatment options for people with COVID-19.
All Māori people aged 50 years and over are eligible for antiviral treatments if they get COVID-19
The number of factors needed to be eligible for these antiviral treatments will reduce, to give greater access for people of younger ages. All Māori people aged 50 years and over, regardless of their vaccination status, will be able to access these treatments without needing other factors for severe illness from COVID-19.
Additional stock of nirmatrelvir with ritonavir (Paxlovid) has been secured
Te Pātaka Whaioranga - Pharmac recently secured an additional 40,000 treatment courses of Paxlovid. It is expected that these treatment courses will be available in New Zealand from late September 2022.
No changes have been made to the distribution arrangements for these treatments at this time. More information about New Zealand’s COVID-19 treatments portfolio, availability of the treatments and how to access them is available on our website.
This change to the criteria significantly expands access to COVID-19 antiviral treatments in New Zealand. If case rates were to significantly increase, we may need to amend the criteria to prioritise available stock for the most high risk people.
Detail about this decision
The Access Criteria
The following Access Criteria will apply to nirmatrelvir with ritonavir (Paxlovid), molnupiravir (Lagevrio) and remdesivir (Veklury) from 14 September 2022. Changes are indicated in bold.
Prescriptions must be endorsed by the prescriber confirming that the patient meets the Access Criteria. For Pharmacist-Only supply, pharmacists must confirm that access criteria are met.
Access criteria – from any relevant practitioner.
Approvals are valid for patients where the prescriber confirms the patient meets the following criteria and has endorsed the prescription accordingly:
All of the following:
- Patient has confirmed (or probable) symptomatic COVID-19, or has symptoms consistent with COVID-19 and is a household contact of a positive case;
AND - Patient’s symptoms started within the last 5 days (if considering nirmatrelvir with ritonavir or molnupiravir) or within the last 7 days (if considering remdesivir);
AND - Patient does not require supplemental oxygen#;
AND - ANY of the following:
- Patient is immunocompromised* and not expected to reliably mount an adequate immune response to COVID-19 vaccination or SARS-CoV-2 infection, regardless of vaccination status; or
- Patient has Down syndrome; or
- Patient has sickle cell disease; or
- Patient has had a previous admission to Critical Care or High Dependency care directly as a result of COVID-19; or
- Patient is aged 65 years or over; or
- Patient is Māori or Pacific ethnicity AND aged 50 years or over; or
- Patient is aged 50 years or over AND has not completed a primary course^ of COVID-19 vaccination; or
- Patient has any combination of three or more high-risk medical conditions for severe illness from COVID-19 identified by Manatū Hauora - Ministry of Health**;
AND
- Not to be used with other COVID-19 antiviral treatments.
Notes:
Consider molnupiravir or remdesivir if nirmatrelvir with ritonavir is unsuitable or unavailable
* As per Manatū Hauora - Ministry of Health criteria of ‘severe immunocompromise’ for third primary dose of COVID-19 vaccine
** People with high risk medical conditions identified by Manatū Hauora - Ministry of Health
^ ‘Primary Course’ defined as receiving at least two courses of vaccination against COVID-19
# Supplemental oxygen to maintain oxygen saturation >93% or at or above baseline for patients with chronic resting hypoxia
How to interpret the access criteria?
We have simplified the criteria to make them easier to interpret.
We have updated the below heat maps to help healthcare professionals interpret the access criteria and identify eligible patients. We will make these heat maps and other resources available on our website, alongside the access criteria, from 14 September 2022.
We will continue to monitor the evidence as it becomes available, as well as available supply, and we will update the criteria as required.
Heat maps to identify eligible patients
Green boxes reflect the updated access criteria from 14 September 2022.
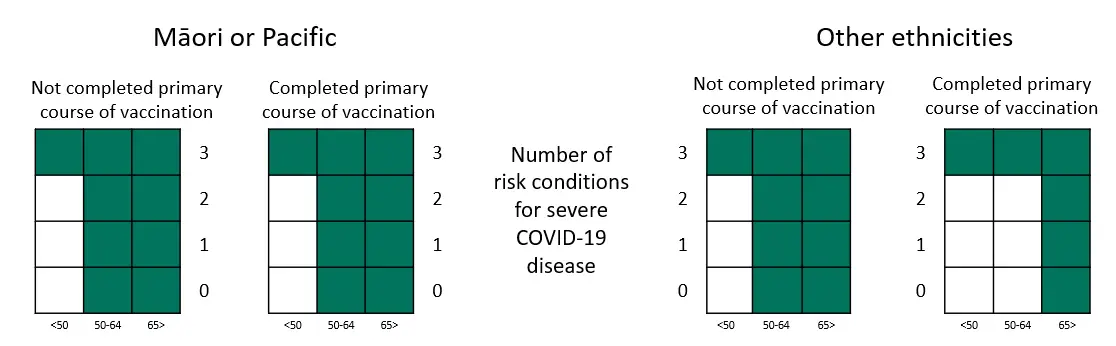
Table representation of the heat maps, showing numbers of high risk medical conditions needed in criteria 4.5 to 4.8
| Age less than 50 | Age between 50 and 64 | Age over 65 | ||
|---|---|---|---|---|
| Māori or Pacific | Completed primary course | 3 | 0 | 0 |
| Not Completed primary course | 3 | 0 | 0 | |
| Other ethnicities | Completed primary course | 3 | 3 | 0 |
| Not Completed primary course | 3 | 0 | 0 |
When people are severely immunocompromised, have Down syndrome, sickle cell disease, or previous critical care or high dependency admission directly for COVID-19 (ie. criteria 4.1 to 4.4), there is no need for any added high risk medical conditions. Below are some example scenarios for eligibility:
Examples are based on the high risk medical conditions listed on the Manatū Hauora - Ministry of Health website as of 12 September 2022. These risk factors may change over time. Please refer to the Manatū Hauora website for the most up-to-date high risk medical conditions.
Example 1: Person is Māori, aged 52 years old and has completed a primary course of vaccination.
This person meets criterion 4.6. If they meet the other criteria, then they are eligible for treatment.
Example 2: Person is of European ethnicity aged 64 years old, has completed a primary course of vaccination and has diabetes, a chronic lung condition, and a BMI over 35.
This person has three risk conditions (as on the Manatū Hauora website) and therefore meets criterion 4.8. If they meet the other criteria, then they are eligible for treatment.
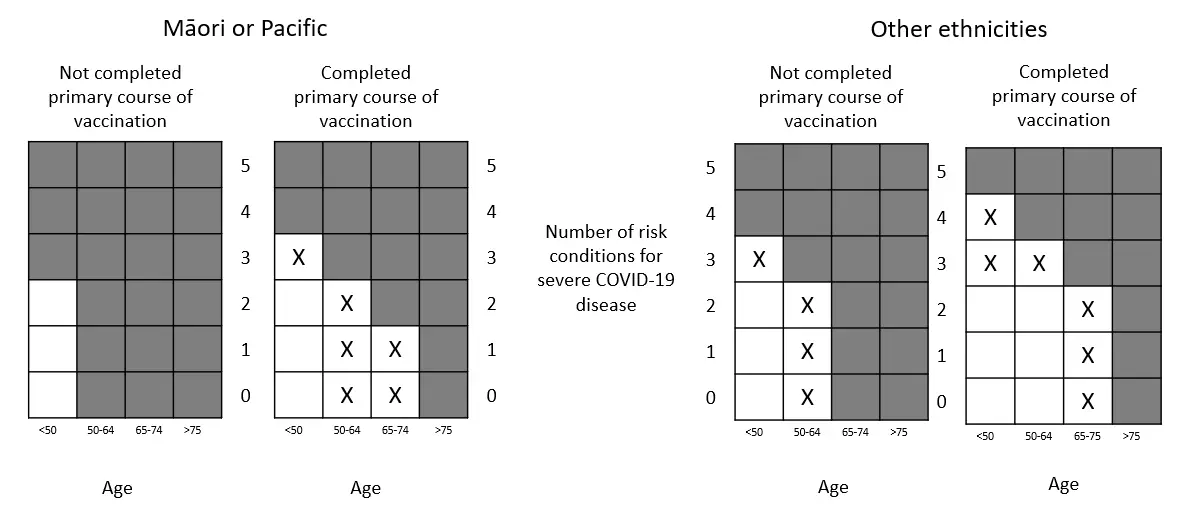
What has changed?
This heat map shows which additional people will now be eligible under criterion 4 of the widened access from 14 September 2022. Grey boxes are the July criteria. Boxes with “x” are the additional groups in the updated access criteria.
Information for people with COVID-19
If you have, or suspect you have COVID-19 and are at high risk of developing severe illness from COVID-19, test early and please get in touch with your healthcare provider. They are best placed to let you know what your treatment options are. Treatments must be started within short timeframes from onset of symptoms. You can find more information on the Manatū Hauora - Ministry of Health website.
Accessing supply of antiviral treatments
Access to antiviral treatments will continue under the current arrangements that are in place.
The antiviral COVID-19 treatments are not accessed via a standard Special Authority. Instead, prescriptions must be endorsed by the prescriber confirming that the patient meets the Access Criteria. The Access Criteria will continue to be available on Te Pātaka Whaioranga - Pharmac’s website and linked to Health Pathways. This approach allows us to easily make changes to the criteria if required in a timely manner.
Antiviral treatments are supplied to pharmacies and Te Whatu Ora - Health New Zealand hospitals at no cost, as they have been purchased directly by Te Pātaka Whaioranga - Pharmac. COVID-19 treatments are funded from a dedicated budget allocated by the Government. That means COVID-19 treatment costs do not come from the annual budget for New Zealand’s medicines (the Combined Pharmaceutical Budget).
Nirmatrelvir with ritonavir and molnupiravir
Nirmatrelvir with ritonavir and molnupiravir will continue to be listed in Section B and Part II Section H of the Pharmaceutical Schedule.
Supply of nirmatrelvir with ritonavir and molnupiravir is currently available to order only by selected community pharmacies and Te Whatu Ora hospitals. This process is being managed by Manatū Hauora - Ministry of Health. More information is available on the Manatū Hauora - Ministry of Health website.
Supply to community pharmacies is currently managed through one wholesaler, ProPharma and Te Whatu Ora hospitals can order from OneLink as required. More deliveries will continue to arrive throughout 2022. The XPharm rule applies to these treatments in the community as Te Pātaka Whaioranga - Pharmac has purchased these medicines directly. Community pharmacies are not able to claim subsidy through normal claiming systems as alternative funding arrangements have been established.
Nirmatrelvir with ritonavir and molnupiravir have been purchased directly by Te Pātaka Whaioranga - Pharmac and there will be no standard dispensing fee or patient co-payment. Pharmacies will be reimbursed by COVID-19 Care in the Community funding through Te Whatu Ora. Information regarding the claiming mechanism for this payment to pharmacies will be available from Te Whatu Ora.
Remdesivir
Remdesivir will continue to be listed in Part II Section H of the Pharmaceutical Schedule.
Supply of remdesivir will continue as it is now and is available to order by Te Whatu Ora hospitals through OneLink. Wholesale stock is held and distributed to Te Whatu Ora hospitals by OneLink in Auckland.
Level 1 hospitals can order and administer remdesivir. Te Whatu Ora hospitals are able to provide remdesivir to both inpatients and outpatients.
Te Whatu Ora hospitals will not be invoiced for supply because this stock has been purchased directly by Te Pātaka Whaioranga - Pharmac.
Where to get more information
For the most up to date information regarding COVID-19 treatments, distribution and stock availability see New Zealand’s COVID-19 treatment portfolio page on our website.
If you have any questions about this decision, you can email us at enquiry@pharmac.govt.nz; or call our toll free number (9 am to 5 pm, Monday to Friday) on 0800 660 050.


